|
7/27/2023 0 Comments Liquid particles Pabst W, Gregorova E, Berthold C (2006) Particle shape and suspension rheology of short-fiber systems. Maranzano BJ, Wagner NJ (2001) The effects of interparticle interactions and particle size on reversible shear thickening: Hard-sphere colloidal dispersions. Transactions of the Society of Rheology 3: 137–152. Krieger IM, Dougherty TJ (1959) A mechanism for non-Newtonian flow in suspensions of rigid spheres. doi: 10.1002/andp.19063240204Įinstein A (1911) Berichtigung zu meiner Arbeit: Eine neue Bestimmung der Moleküldimensionen. International Journal of Multiphase Flow 1: 195–341. doi: 10.1017/S002211207700106īrenner H (1974) Rheology of a dilute suspension of axisymmetric Brownian particles. 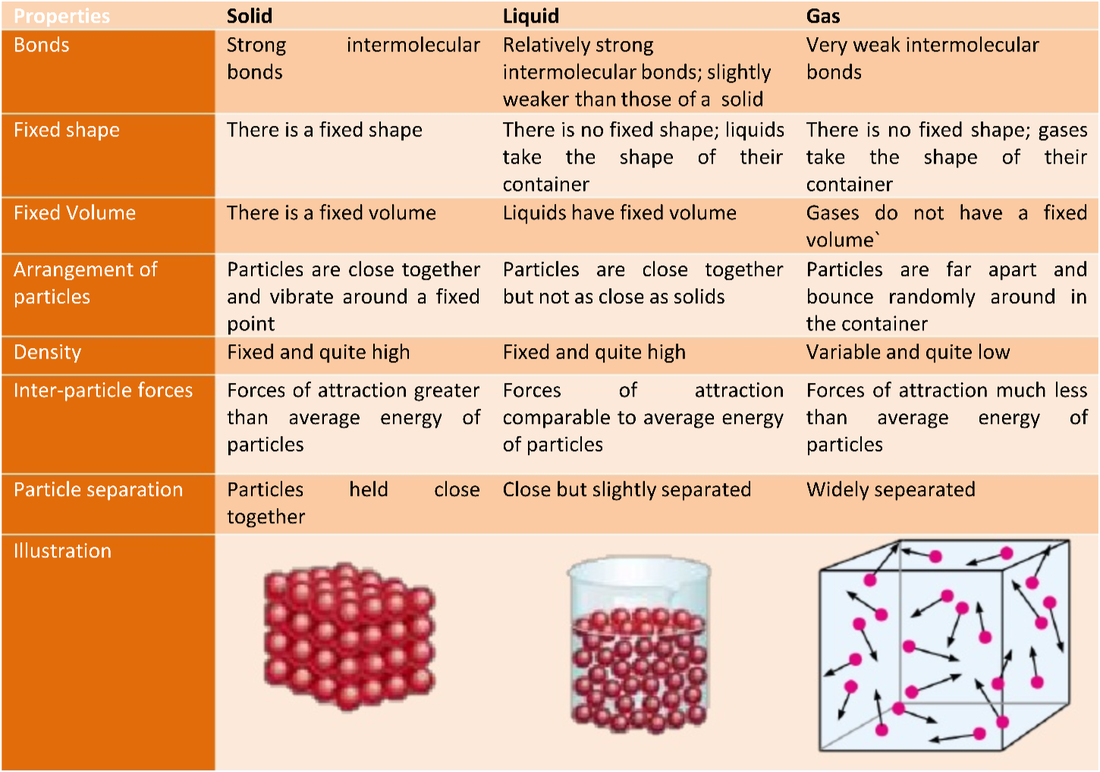
doi: 10.1122/1.1343879īatchelor KG (1977) The effect of Brownian motion on the bulk stress in a suspension of spherical particles. doi: 10.1122/1.1413504Īncey C, Jorrot H (2001) Yield stress for particle suspensions within a clay dispersion. Except for dynamic light scattering (only applicable to wet samples), these methods can be used for both dry and wet samples.Īncey C (2001) Role of lubricated contacts in concentrated polydisperse suspensions. This method is particularly useful if particles show strong deviation from sphericity. Several thousand images are taken to ensure statistically sound values. The obtained values can be converted to zeta potential distribution or an average zeta potential, respectively.Īutomated imaging allows determination of particle shape and particle size if particles are at least 1 µm in diameter. Charged particles will now move towards either the positive or negative electrode, the velocity is measured with the laser Doppler technique as frequency shift or as phase shift. Hence, the particle speed can be correlated with particle size.įor electrophoretic light scattering, two electrodes are connected to the sample and an electrical field is applied. The measuring principle uses intensity fluctuations over very short timescales due to Brownian motion. However, the measurement is based on the assumption of spherical particles and knowledge of the optical properties of the solvent as well as the particles is required.ĭynamic light scattering allows particle size measurement down to the sub-nm range. The light scattering behavior differs with respect to the particle size: smaller particles scatter more strongly than larger particles. The measuring principle is based on having a laser directed to the sample and multiple detectors to record scattering at different angles. Laser diffraction is able to analyze particles from the sub-µm to the mm size range. Furthermore, electrophoretic light scattering is used to determine the zeta potential. These properties require additional methods like dynamic light scattering or laser diffraction (for particle size and PSD) and high-resolution imaging (also particle shape) to be measured. Characterization of the solid fraction requires knowledge of particle size, particle size distribution (PSD), and particle shape. This article aims to separate the effects of particle concentration, particle shape, particle size distribution, and particle surface charge, and to name common methods to measure these properties.īulk physical properties of the suspension and the solvent, like viscosity and yield point, can be measured with viscometers or rheometers in rotational or oscillatory tests. Furthermore, the particle surface’s electrical charge (zeta potential) influences the suspension’s rheological behavior. In most suspensions, particles differ in size, which can be expressed as the particle size distribution. 
However, the materials’ viscoelasticity not only depends on the concentration of particles but also on particle shape and particle size. A low particle volume fraction (termed “solid fraction” hereafter) typically induces shear-thinning behavior, whereas high particle concentrations might result in shear thickening. Many liquid products, paints, inks, beverages, medicine, slurries, or shower gels contain various types of particles to achieve the desired final product or adjust processing properties. In case the particles are insoluble, the product is a two-phase (or multi-phase) mixture which is termed a suspension if particles are larger than 1 µm or termed a colloid if particles are between 1 nm and 1 µm in size. Particles are dissolved as molecules or ions (<1 nm). If the particles are soluble in the solvent, the final product is termed a solution. 
Solvents, such as water or acetone, are typically ideally viscous liquids, while particle-bearing liquids display a more complex rheological behavior. Adding particles to liquids modifies the liquids’ optical and physical properties, e.g. 
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
